At mid-year, it has been a custom here at Eye on FDA to look back at a few categories of activity by FDA at the first half of the year and discern what it tells us about future directions. This year the unprecedented circumstances presented by the COVID-19 pandemic make it particularly interesting this year. A good place to start is with regulatory enforcement of the Office of Prescription Drug Promotion (#OPDP).
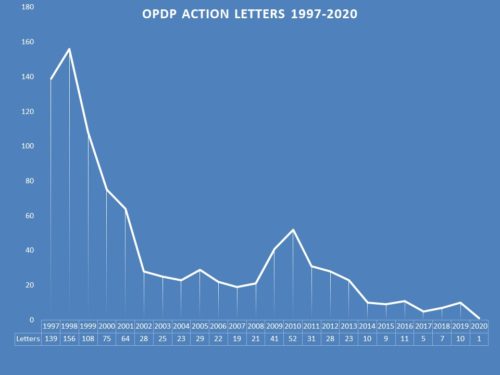
In the now distant past, enforcement from this office, then called the Division of Drug Marketing, Advertising, and Communications (DDMAC) and later re-named OPDP, was robust, with the office issuing scores of letters a year (156 were issued in 1998). However that number begin to trend downward and in the past several years, the number of letters issued has dwindled ranging from as few as 5 to as many as 11 over the past five year period.
There are potentially many reasons for this drop in enforcement, which could be the subject matter of its own for a blog posting. But this year, enforcement is particularly lull. There has been a single letter (1) issued this year, issued in the nascent days of the pandemic on February 21.
The interesting thing in connection with that is that in May – in the midst of the COVID-19 pandemic – FDA held a Webinar on the “Bad Ad” Program – a program that deputizes healthcare professionals to assess promotional communications and report potential violations to the agency. By holding the Webinar, OPDP is presumably to solicit greater participation of healthcare professionals but in spite of the fact that visible regulatory enforcement is at an all time low. But truly, one has to wonder in the midst of a pandemic that is gripping and challenging the healthcare system just how much spare time healthcare professionals might have to participate in a non-reimbursed activity in this regard. Moreover, how much of a priority is enforcement given the track record of the past several years?
This year’s single letter was a Warning Letter, the more serious type of regulatory action letter issued by OPDP. The communications vehicle that incurred the violation was a sponsored link that appeared on Google. The drug in question is Schedule II drug and bears a Boxed Warning on its label, yet the link contained no risk information about the drug, despite the fact that there was present information about the benefits of the drug, according to OPDP’s letter.
OPDP has indicated in the past that the use of sponsored links without risk information is a no-no. Notably in April 2009 the agency issued 14 letters regarding 45 brands all related to the use of sponsored links – going after a practice known as the so-called “one-click rule” which was not a rule at all, but the common practice in promotional communications of having risk information just one-click away in a promotional communication. The agency had never said that this was not a permitted practice and only made it clear by issuing the letters, rather than having enunciated the parameter through a guidance document about digital communications.
Then as part of the agency’s effort at putting together a regulatory stance regarding the unique questions posed by the huge migration into digital and social communications, FDA later that year posed a series of five questions to inform the regulation of this burgeoning environment during a public meeting held that same year. One of them was about the appropriate use of links. FDA said they would produce a guidance by the end of 2010, but did not. Only in 2014 did the agency issue guidance documents addressing some of the questions they posed in their framework, but have not issued anything since and have left many questions unanswered while new ones have emerged. For some years CDER published its Guidance Agenda to include a guidance on the use of links to third party websites by pharmaceutical companies. This item disappeared from the agenda, however, and no draft guidance was issued.
As we muddle our way through the COVID-19 pandemic, one of the emergent truths is that virtual and digital life is taking on a much greater presence than is real life. We are on Zoom calls, FaceTime cocktails and our use of the Internet has apparently surged. Utilization of the services of physicians and other healthcare providers, on the other hand, have plummeted. It stands to reason therefore that use of the Internet for health-seeking information is likely on the rise during this time. Telemedicine is ascending and patients are going to be relying on what they access online over what they access in a doctor’s office, or from television for that matter.
Yet the focus of OPDP seems to continue to be on traditional communications vehicle, while leaving the work on digital and social undone. Recently, FDA published in the Federal Register intent to study the promotion of multiple indications in Direct-to-Consumer television advertisements. In fact, the OPDP research agenda has a focus that is heavily tilted to traditional avenues of communication over digital and social, despite the fact that it is the latter to which people are turning for healthcare information. Several aspects of digital and social guidance are in need of attention – from designing a website for mobile use to the appropriate use of links. The pandemic certainly presents one not only with the opportunity to provide clarity on these and other aspects, but the overwhelming need for such attention. It is difficult to say what enforcement will look like for the balance of the year but it certainly seems like a good time to focus on some gaps.

