
Each year, midway through it all, I take a look back to see what FDA has been talking about and compare it to the year before to see how things may have changed. I’m a little late at getting to it this year due to all the COVID-19 distraction. But it is not likely to surprise anyone that the first half of 2020 has caused not only an increase in communication from FDA, but a focus as well.
During the first half of the year, FDA issued a record number of press releases and nearly two-thirds (64 percent) were about COVID-19. The first release related to the pandemic appeared on February 24 to inform us about what steps the agency would be taking with respect to foreign inspections. FDA acted early and relatively often after that. The agency now issues daily updates on the topic.
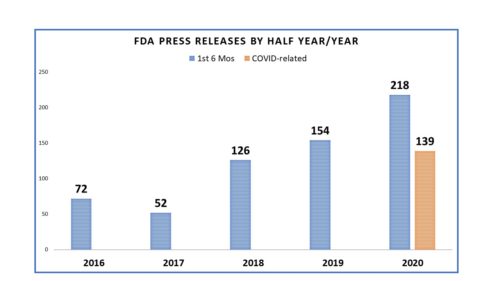
Let’s first examine the profile of all the releases, and then focus on those just about COVID-19.
Approvals – There were 54 press statements regarding approvals so far in 2020, comparing to 42 approvals announced in 2019. The big difference, however, is that a big slice of approvals in 2020 involved Emergency Use Authorizations (EUAs) for COVID related products. In fact, there were 16 EUA announcements in 2020, which if you subtract that from the total of 54, leaves you with 38 non-COVID announcements which would lag just a bit behind last year.
The breakdown of those approvals between drugs and devices is also skewed this year due to the fact that the overwhelming number of EUAs about which statements were issued involved devices – usually diagnostic testing associated with COVID. Fully 15 out of the total of 16 EUA announcements involved devices – with only one involving a drug announcement (the EUA for remdesevir). So for the total number of approvals, including EUAs in 2020, the split among the total of 54 approval announcements in2020 were 33 drugs and 21 devices, comparing to 2019 when there were 29 drug approval announcements, 12 devices and 1 gene therapy. If you back out the COVID-related EUAs for 2020, it would be 32 drug approval announcements with only 9 device approvals for 2020, which would have it behind last year at this point.
Alerts – Normally there are not too many alerts in a given year, but 2020 has been an alarming year to say the least. The agency sent out 9 alerts during the first half of the year, 4 of which were related to COVID-19, including one for the off-label use of anti-malaria medications. During 2019, the agency had issued 10 alerts related releases. And so the impact of COVID-19 on alerts did not increase the volume, but were involved in the subject matter of almost half of them.
Guidance/Policy Announcements – There has been more guidance from the agency this year, again due to COVID-19. During 2020, the agency issued 13 releases involving guidance announcements or policy changes, 11 of which were related to COVID-19 compared to the first half of the previous year when there were only 5 Guidance document-related releases. The non-COVID related guidance for 2020 was in relation to e-cigarettes and were issued during the first quarter. Everything subsequent was COVID related, indicating how the pandemic has consumed the focus of the agency.
Legal – These announcements include warnings to companies and court actions such as consent decrees. During the first half of the year, FDA issued releases about 14 legal actions, somewhat less than last year when it issued 19. Of the 14 issued this year, half of them were COVID related in some way, including the announcement regarding the agency’s revocation of the EUA for use of hydroxychloroquine as a treatment for COVID. A sidenote – it does not appear the agency issued a release regarding the issuance of the EUA for hydroxychloroquine. The legal announcements were almost entirely centered on drug related products, with 3 related to tobacco and only 1 for a device.
General Announcements – This is the largest category and the place where “other” goes, i.e. anything not in the above categories. It is the place for agency updates on various topics and providing information that is not specific to an agency action. It includes statements from the commissioner and various division heads. During the first six months of the year, FDA issued 51 general announcements, 30 of which were related to COVID. These covered subjects ranging from calls for blood donations to updates on policy developments to statements regarding the passage of legislation. This compares to 114 that were issued during 2019. While that would seem to be a lot fewer, in fact there has been a real diversion to talking about COVID. In addition to these general announcements, FDA has issued 72 Coronavirus Daily Updates which carry a miscellany of compiled updates on the agency’s activities that are not circulated via a stand-alone press release.
In short, the volume of what is coming out of FDA has increased significantly. This happened when Dr. Gottlieb took over the agency and began handing out Commissioner Statements like crazy. It has happened again because public health circumstances. But there is more to communications than volume, and in an upcoming posting, I’ll take a look at content and assess how well FDA is doing with regard to the content.
Photo by AbsolutVision on Unsplash

