
Looking back at FDA Advisory Committee meetings (AdComms) held during any given year requires a bit of patience given that some of the approval results don’t come in for a while after each meeting is held. And in fact, there are still some drugs that were considered by advisory committees during 2018 for which there is still no FDA decision. But we are going ahead with a look-back anyway.
FDA holds advisory committee meetings in order to get input from experts and from the public regarding the approval of new products and has a fleet of advisors serving on those committees. For Human Drugs there are 18 committees with varied subject matter jurisdiction. Not every new drug application goes through the advisory committee process, but for those that do, from a communications perspective, it is often where the branding of a medicine takes its first breath.
That is because when there is an advisory committee meeting held it does bring more transparency to the approval process because there is a publicly open scrutiny of the application. This, in turn, has communications ramifications. While the application milestones and clinical study outcomes have largely been public, the bulk of attention has been paid in trade and scientific journals. When there is an AdComm, there is a shift to mainstream media coverage. And while the filing of the new drug application and the acceptance of that application are important milestones, it is when FDA posts the briefs of the professional reviewers within FDA that result in headlines in mainstream media. And because reviewers are citing their perceptions of the strengths and weaknesses of the drug in question, the comments in those briefing documents generate the first real round of headlines about the medicine. The second wave comes as a result of the meeting discussion and vote. And as we know, the vote is a recommendation to FDA which the agency may or may not follow.
All that said, how did 2018 stack up in terms of Advisory Committee activity and how did it compare to the year before? Last year, FDA approved a record number of new molecular entities (59). In addition there were approvals of biosimilars and approvals of drugs that were not new molecular entities, as well as label expansions.
More Meetings. There were more meetings held last year than the year before. In all there were 34 meetings of advisory committees last year, but only 27 of them involved the consideration of New Drug Applications (NDAs) or supplemental New Drug Applications (sNDAs). That is slightly higher than last year when there were 29 meetings, 23 of which were considering NDAs for approval, two of which ended up not taking place.
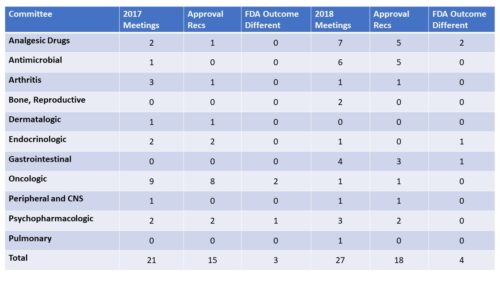
Most New Treatments Considered. In 2017, the Oncologic Drugs Advisory Committee came in at a whopping 9 meetings to consider drugs, but in 2018 met only once. The committee with the most meetings in 2018 was the Analgesic Drugs Advisory Committee with 7 meetings, indicative of the activity around pain. Also in 2018, the Antimicrobial Drugs Advisory Committee met 6 times.
Voting Patterns/No Votes. In 2017, the Committee that had the highest number and proportion of negative outcomes was the Arthritis Drugs Advisory Committee, voting down 2 out of 3 NDAs, while in 2018, it was the Analgesic Drugs Advisory Committee that voted down 2 out of 7. In 2017, the Advisory Committees voted to recommend approval 71.4 percent of the time; in 2018 it was 66.6 percent.
Disagreement in Outcomes Between FDA and AdComm. As is noted in every press release about an AdComm vote, FDA may or may not follow the recommendation of the committee. That naturally raises the question – how often does FDA go against the advice of an advisory committee. Consulting my own database that tracks outcomes, I have found that FDA goes against the advice of the committee about 11 percent of the time. In 2017, there were 21 recommendations and FDA went counter to the recommendation of the committee in 3 of them (14 percent). In 2018 there were 27 votes and FDA appears to have gone against the recommendation 4 times (15 percent). There is still one FDA decision still pending where FDA has extended the PDUFA date for the investigative treatment for postpartum depression Zulresso(TM) from Sage Therapeutics. If FDA’s decision ran counter to the AdComm recommendation of approval, would change these numbers slightly.
For your reference here is a table comparing the activity of the past two years. The first column denotes the number of meetings/medicines considered by each committee, the second the number of approvals voted, the third the number of times FDA had a different outcome from the recommendation.


Pingback: Recon: Merck Acquires Immune Design for $300M; CMS Projects US Health Spending to Rise 5.5% Annually Over the Next Decade – Regulatory Focus | Everyday News Update