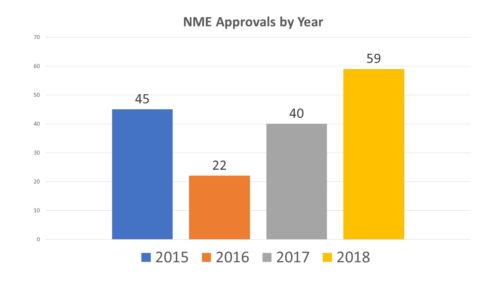
Last year new molecular entities (NMEs) were approved by FDA at a record clip, surpassing the prior record of 45 that was set in 2015. As we begin the final quarter of hte year, this is a good time to check in on how we are faring in 2019.
It may have been tempting to attribute last year’s spike in approvals to the passage of the 21st Century Cures Act (the Cures Act), pointedly designed to facilitate and speed up innovation and new drug, biotech and device approvals. But the implementation of an Act as broad and deep in scope as the Cures Act takes time. Any credit to the Cures Act for the increase in NME approvals may have been premature.
In a mid-year check up posting for this year, it was noted that approvals of NMEs at FDA was running far below the pace at which it was the previous year and that the number of known PDUFA dates for NDAs and BLAs (versus sNDAs and sBLAs) appeared to be lacking in number to make this year equal or surpass last. With less than three months to go before 2020, the news from the third quarter is that nothing has changed. Comparing approvals by the end of the third quarter of each year, one can see that we are in line with 2015. That would not seem to rule out the fact that we could still have a good year.
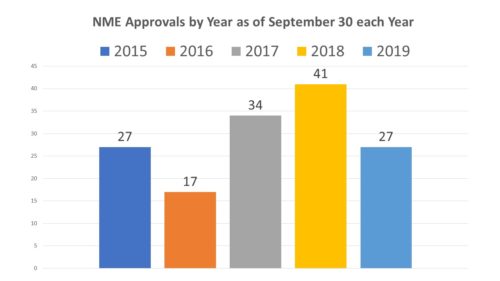
At the mid-year mark earlier this year we were at 17 NME approvals. Having added 10 new approvals in this quarter, the pace has certainly picked up. An approval rate like that for the last quarter would certainly bring this year into line with 2017 and 2015, but still far short of last year. What does the 4th quarter look like?
That leads one to look at known PDUFA dates that are upcoming to see if there is a well upon which to draw for future NME approvals this year. While proprietary, I track PDUFA dates in a database as they become discoverable or can be estimated from public information. By my count there are 33 dates for this quarter for FDA decisions on submissions, which suggests a big potential for approvals of NMEs. But two things – first not all submissions are for NMEs, thereby narrowing down the possibilities. The second thing is that of those 33 dates, only 18 are for NDAs or BLAs, and only 4 are priority reviews, suggesting that there is perhaps not a deep well of NMEs available.
Time will, of course, tell. But as one attempts to interpret the true impact of the 21st Century Cures Act, the fact is we may have to wait a bit longer to realistically assess its benefits, at least as far as new approvals and innovation are concerned.

