
Happy New Year! It is 2020 – the year of clear vision. Let’s look back for a bit as we plunge forward.
FDA is a giant agency where a lot happens. Narrowing down the actions of significance is therefore a very subjective experience. Even limiting the subject matter to those actions which affect medicine, as opposed to food, devices and cosmetics, there is still a very wide bandwidth of activity and impact. And everyone gets to have their point of view. Having been involved in writing this blog since 2006, here is mine. Some of these noted items are things FDA has done – great milestones and achievements – some involve things the agency has not done – and others may involve situations that are evolving around the agency.
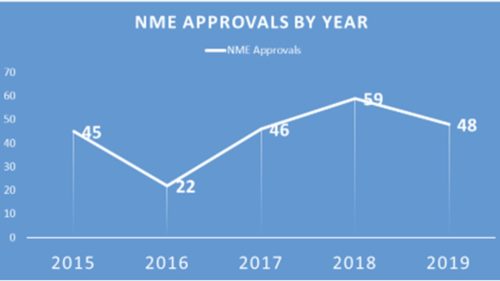
- Fewer NMEs, But Interesting Approvals – At 48, there were fewer New Molecular Entities (NME’s) approved in 2019 than in the previous year when there were a whopping 59, but it was still more than in 2017, 2016 and 2015, thanks to a spate of approvals in December. That said, there were a number of interesting developments within the greater span of approvals, including some NMEs. These included the approval of two new treatments for Sickle Cell Disease, the approval of the first Gene Therapy (and incidentally the most expensive drug to be on the market); the approval of the third cancer treatment based on the presence of a genetic characteristic rather than tumor type; the approval of two new anti-depressants in a category that has long been without advance; and in vaccines, the approval of the first preventive for dengue as well as one for Ebola and the first test to detect Zika virus antibodies.
- Three Commissioners in One Year – FDA started the year with Dr. Scott Gottlieb at the helm who surprised everyone with a departure in the Spring. In turn, he was replaced by the solid choice of Dr. Ned Sharpless who worked as Acting Commissioner for most of the year. Another surprise, despite the support of many past FDA commissioners, Dr. Sharpless was not appointed FDA Commissioner. Rather Dr. Stephen Hahn was sworn in as the 24th Commissioner of FDA. How the advent of a new commissioner will impact policy is anyone’s guess. During the confirmation process there was some question as to the commitment he has on the FDA’s efforts vis a vis the vaping industry, but as we learned during the Gottlieb confirmation process, concerns raised before don’t always materialize as deficits after confirmation. Even with the advent of a new commissioner, expect the media shadow of Dr. Gottlieb to extend to future FDA activities and policy announcements.
- FDA Moves on Importation – Apart from generics, this is the other area where the agency can contribute to the effort to bring down the price of prescription drugs. It was an historic first in many respects – the first time the agency facilitated the pathway for the importation of drugs by issuing a draft guidance document and a rule. The former sets up a regulatory pathway for states and other entities to import drugs from Canada and the latter devises a means by which manufacturers can import drugs from outside the U.S. in order to lower the price in the U.S. though it remains a mystery as to why any manufacturer would likely do such a thing. The other historic thing about the announcement from FDA was that it was the second time the agency politicized the policy announcement by linking it to the Administration – the first being in September. For thoughts on that see the previous posting.
- CBD Enforcement – CBD is everywhere. Everywhere geographically, everywhere in the span of products – teas, salves, dummies, hand cream, vape pens – you name it, CBD is likely there leading many stakeholders to label it the “Wild West” to signify that it is without oversight. In the wake of the Farm Bill which made legal the growth of hemp, FDA issued a press release in an attempt to preserve the agency’s regulatory authority over its use as an ingredient in supplements, drugs or food. That was despite the fact that the market was already running amok with products containing CBD. After his departure from FDA, Dr. Gottlieb penned an op-ed in The Washington Post outlining what FDA needed to do as the use of CBD was “getting out of hand.” During the course of the year, FDA issued a spate of warning letters to multiple manufacturers of a range of products including food producers warning them against the use of CBD. For an overview of FDA activity on this front, see “CBD and FDA – Where Are We?” Since that posting, FDA has issued additional letters. Can FDA send enough such letters to actually chill the CBD-related product market? Certainly the agency is under a measure of scrutiny given the opioid and vaping epidemics that are also high profile.
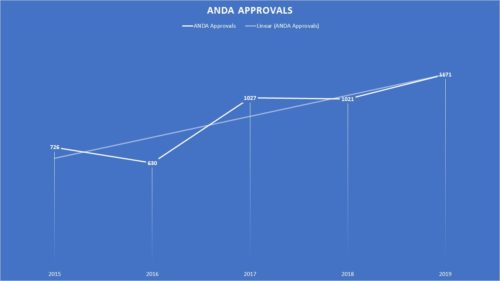
- Record Number of Generic Approvals – The agency approved a record number of new generic drugs – one of the few contributions that FDA can make to impact the price of pharmaceuticals. The agency approved 1171 new drugs during the fiscal year. This follows on the heels of two notable years of approvals in 2017 and 2018 Notable announcements included the approval of a generic version of Advair Diskus, valsartan, naloxone nasal spray to treat opioid overdose, Gilenya – an oral treatment for multiple sclerosis, and the approval of a generic Eliquis in the atrial fibrillation treatment space. In addition, FDA issued multiple press releases during the course of the year providing updates on its progress to enhance the approval of generic drugs. With the backlog clearing out, it will remain to be seen if the pace continues in the new year.
And for 2020? Look for a likely uptick in attention to gene therapies as experience with those approved becomes more apparent and those in the pipeline get closer to the market. Watch what happens on the e-cigarette front given the concerns on the topic raised at the time of the new commissioner’s confirmation and monitor how FDA responds to the most recent criticisms of how the agency has handled the opioid epidemic. Probably best not to look for any further guidance on social media and communications by pharma as most of the research directed by OPDP is looking to traditional communications vehicles. As patients and the public have moved into social and digital for media consumption, OPDP remains with its head stuck in the sands of broadcast DTC.
Photo by Andrea Davis on Unsplash

