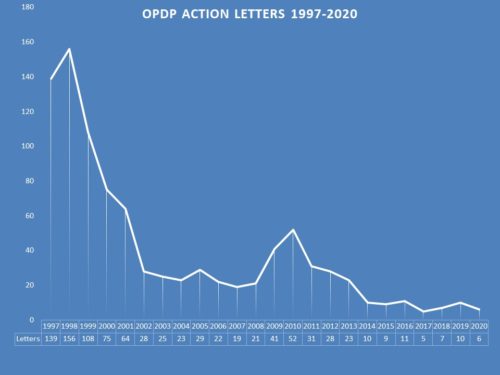
Last month we looked at a review of FDA press releases for last year and as one might have expected, there was an enormous spike in communications as a result of the COVID-19 pandemic. Looking now to a review of enforcement by the Office of Prescription Drug Promotion (OPDP) in 2020, we see a different picture. We instead see the same pace as we have the past several years – that is anemic levels of enforcement. And as in recent years, the regulatory action letters were exclusively aimed at smaller, less experienced companies. Whether that is by design, or by virtue of the fact that they are more prone to regulatory error, is not clear. The collection of letters issued by OPDP during 2020 can be found here.
Characteristics of Enforcement for 2020 – One thing that stands out this year was the fact that the bulk of the letters issued to companies regarding violations were Warning Letters rather than Untitled Letters. Warning Letters are the more serious of the two types of letters sent by the agency. So when OPDP did act, they meant business. Let’s look to the other characteristics of the letters for 2020:
- Five of the six letters were Warning Letters rather than the less serious Untitled Letter
- Five of the letters involved products with Boxed Warnings
- Four of the letters involved digital communications, two sponsored links and two emails were the subject of action
- Two were focused on traditional direct-to-consumer ads – one radio and one television
- Half of the letters were prompted through the Bad Ad Program (see below)
The Violations – In recent years, there has been an uptick of activity aimed at the promotion of an unapproved drugs. While this year did not see such a violation, there was a violation related to promotion of an unapproved use. In addition, the omission or limitation of risk information – the most common subject matter of a regulatory action letter by FDA, was represented in each of the letters issued during 2020. In all there were 6 letters containing 13 violations – 6 for risk; 1 for an unsubstantiated claim; 2 for broadening indication; 1 for unapproved use and 3 in the “other” category which included a failure to use the established name in the promotion.
- All of the letters involved risk minimization or omission
- A reminder that risk information cannot be incorporated by link to “full prescribing information” or any other source
- Risk information cannot be cherry-picked, particularly from a Boxed Warning
- One of the letters involved a promotion that did include the boxed warning in the communication, but lacked other risk information and placed the risk information out of proximity to the statements of benefit – Presence of risk information alone is not adequate, but rather the presentation must be balanced
- One letter included a violation for promotion of an unapproved use where there was a suggestion that the compound relieved respiratory symptoms associated with COVID-19
Heightened Role for “Bad Ad Program” – A recent interview with the new Acting Director of OPDP revealed that a primary focus for the office this past year was in relation to the biosimilars market. It was also noted that the “Bad Ad” program turned 10 years old this year. The Bad Ad program, for those new to this, was instituted to deputize healthcare professionals in the role of helping to referee when promotional communications are perceived to go outside regulatory parameters. Notably, it appears that at least half of the six regulatory action letters issued this year were prompted through Bad Ad program reporting. The article states that the program had received over 2000 reports over its lifetime. Since there have been no where near that number of enforcement actions by OPDP during that time, it appears that there is a lot to sift through.
Social and Digital Work Left Undone – With regard to social and digital media, the Acting Director echoed the sentiments of the past when saying that FDA is focused more on the message than the medium. This is despite the fact that the guidance work that was put into motion in April 2009 framework set up during the public meetings about social media remains partially undone. There are still distinct questions relatable to digital and social media which have never been addressed which are platform specific – such as optimization of a branded web site for mobile, the responsible use of links, and adverse event reporting. CDER Guidance Agendas in the past had included some of these topics, but are no longer included in the recent agendas and appear to have been dropped.
One resource to examine when considering the potential directions for OPDP enforcement and guidance development is the identified “Research in Progress” which the office is undertaking that often informs future direction. If interested to see what they are looking at in terms of digital and social, you will be disappointed. The agenda still has a focus on DTC print and television ads. Nothing about risk presentation on mobile sites.
Conclusion – Finally, in the OPDP interview referenced above, it was indicated that a priority for the coming year will be to make the work of the office more visible and relatable, as well as to continue vigilance for claims being made around COVID-19 treatments. Overall, it would seem that despite the change in leadership, priorities remain the same – reviewing launch materials for new drugs and focusing on enforcement with respect to specific categories where there is high risk (pain medications) and situations where there is a greater risk as represented by the presence of a boxed warning. In other words, they are concentrating their resources on the greatest risk areas rather than spreading it out across the spectrum as was done when the number of regulatory action letters in a year far exceeded the current output.
For 2021? So far, no letters. Last year, the first regulatory action by OPDP did not occur until March. Stay tuned.

