
It has been a regular feature of the blog to give a read on what FDA has been talking about, at least through the form of press releases. It is not always an easy task because the nature of FDA’s practice in this respect has evolved over time. But here is what we see looking at the first 6 months 2021, and comparing it to mid-years past.
You may recall from the last posting on this topic at the beginning of the year, during 2020, FDA had a lot to say – a real lot, and that isn’t surprising given the agency’s role in facilitating treatments, diagnostics and vaccines to respond to COVID. Last year the agency issued over 400 releases on a range of topics (65 percent of releases were related to COVID-190. And while we are still in the midst of the pandemic, it is somewhat surprising to see that the overall number of releases during 2021 has fallen off a bit – to 153 so far this year (54 percent have been COVID-related), which if this pace kept up, would annualize to only about 300. Of the 153, only 33 of them were translated into Spanish.
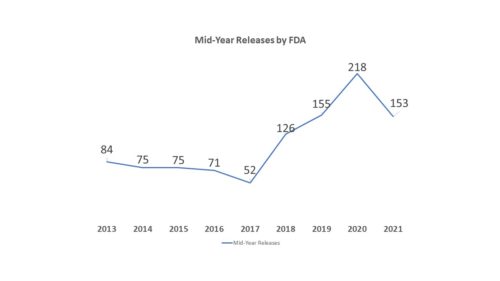
It is even more surprising considering that the agency started listing releases under “FDA in Brief” in with its listing of Press Release materials, which serves to inflate the total number of press releases. Consider “FDA in Brief” releases as a sort of press release-lite. They have their own section in the FDA Newsroom and usually involve topics that are not quite press release-worthy by my observation. They generally have a more limited audience and a different format than FDA press releases. And while some, but not all, of FDA’s press releases are also issued in Spanish, FDA in Brief releases do not seem to ever be available in Spanish. It would appear in May of this year, the agency began adding “FDA in Brief” releases into the list of regular press releases, while still listing them separately under their own heading, adding about 20 entries. So what that means, if you backed those out is that the agency actually only put out about 133 press releases by mid-year, compared to last year when FDA put out 218 press releases.
Also by mid-year last year, FDA announced 55 approvals, including several Emergency Use Authorization (EUA) announcements for drugs and devices compared to just 44 approvals, EUA’s, or label expansions so far this year (though the number of new molecular entities is running at a very high pace during the first half of this year). This year 10 of these approval announcements have been in relation to COVID-related medicines or devices, while last year it was 17.
Further, last year FDA introduced regular Coronavirus Updates as part of the press release regimen. Last year during the first half of the year there were 134 such updates. This year there were only 67 Coronavirus Updates in the first half.
With regard to other subject matter, there were 6 alerts issued by the agency during the first half of the year and 9 releases regarding legal actions including seizures, consent decrees and warnings.
There is no telling for the reason or reasons there has been a drop-off. Certainly last year may have been an anomaly given the circumstances. But this year there were also fewer releases by mid-year than in 2019 when there were 155, though on par with the mid-year level in 2018 when there were 126. Any number of factors could be affecting the agency’s output, including the fact that the agency is still working with an Acting Commissioner. As we saw with the “Gottlieb effect”, Commissioners can make a difference in the agency’s approach to communications. We’ll check in again at year end.
Photo by AbsolutVision on Unsplash
Author’s Note: Unfortunately the first automated mailing to subscribers last week did not work out so well and subscribers received a blank email when they should have received a blog posting about an Untitled Letter issued by FDA’s OPDP. Apologies for the misfire, but hopefully we are working more smoothly now. Thanks!

