
In January we took a look at FDA Advisory Committees (AdComms) – FDA AdComms – When the Going Gets Tough – noting, among other things that there had not only been fewer meetings but also that it appeared that the outcomes were increasingly negative.
Just to recap, for the entire year of 2022 there were only 14 meetings held to consider whether new medicines should be recommended for approval, a slight increase over the 10 the year before, but considerably lower fewer than 2018 and 2019 when there were 29 and 22 meetings held, respectively. And as can be seen by the increase in red bars (negative outcomes) and the decrease in positive outcomes (green bars), the outcomes have skewed increasingly negative, with 2022 being a particularly negative year. Note these numbers cover meetings about drug applications only, not including vaccines or cellular therapies.
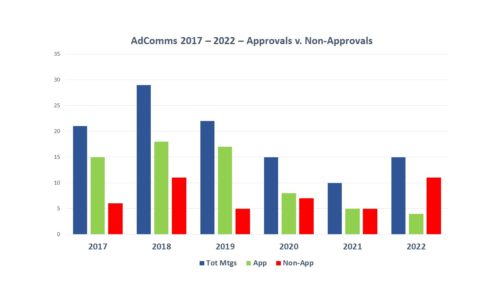
By contrast, already in 2023 there have been eight AdComm meetings scheduled by FDA for the purpose of considering a new medical treatment, three of which have occurred and all resulting in votes in favor of the sponsor, so perhaps 2023 is heading to less negative territory. During the balance of this month and into next, there are five more meetings scheduled, so we shall have to see.
That tells the collective story, but how do each of the committees fare?
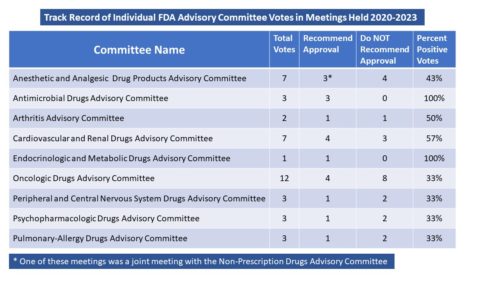
The committee with the most AdComm meetings was also one of the committees with the highest number of negative outcomes – the Oncologic Drugs Advisory Committee. The committee met a dozen times during 2022 and only voted in favor of recommending approval four times. Two committees – the Endocrinologic and Metabolic Drugs Advisory Committee and the Antimicrobial Drugs Advisory Committee were among those that met the fewest times – once and three times, respectively, but both had purely positive outcomes. In fact, bottom line, there were nine different committees that had meetings to consider new drug approvals last year, only four of them voted positively 50% or more of the time, with five of them voting negatively more often.
There are a lot of variables that factor in. Drugs for cancer face a whole different set of criteria for assessing safety and efficacy than do other therapeutic categories. That said, beginning in 2020 meetings began skewing to more negative outcomes and 2022 was the first year where the number of negative outcomes outweighed the number of positive outcomes. As noted earlier, this coincides with the transition to virtual meetings over in-person ones.
But the nature of AdComms may be in flux calling into some question the role and influence of voting. Recently, Commissioner Califf was quoted in a few articles this month reporting on a moderated discussion in which he participated at the Biopharma Congress meeting held in Washington, D.C. in mid-February. The coverage in part focused on his thinking that advisory committees need to be less aimed at actual votes rather than on having a full discussion of the issues. Apparently these comments did not come in the form of a speech which would be listed on the FDA’s website which might provide some context – because on their own, they are rather confusing. More thinking about this in an upcoming posting, but it signals perhaps that the nature of AdComms may begin to skew less to circumstances where there are voting questions posed to the committee and that there may emerge a more consistent use of “discussion” points instead. If so, that will certainly complicate future big picture analyses, forcing the discussion to be more qualitatively than quantitatively focused.

